Apple reportedly secured regulatory approval from the US Food and Drug Administration (FDA) for its new hypertension detection feature, set to launch next week.
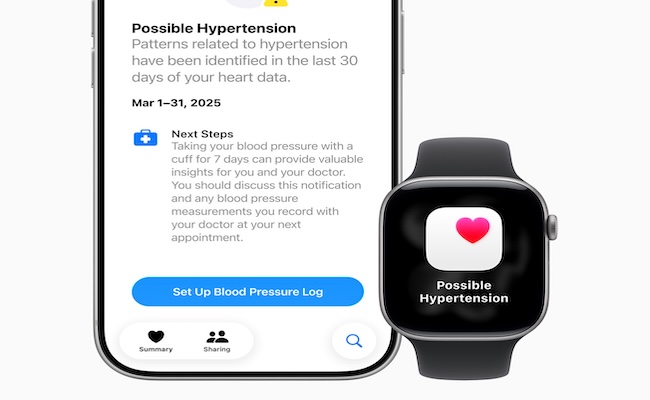
First introduced during the device giant’s product launch event earlier this week, the feature utilises the Apple Watch’s optical heart sensor to monitor how a user’s blood vessels respond to heartbeats. The algorithm operates passively in the background, assessing data over a 30-day period. If consistent signs of high blood pressure are detected, users are alerted and encouraged to consult a healthcare provider.
Previously, the company said it was awaiting clearance from the FDA and other regulatory bodies before launching the feature globally. Following the green light, the tool will now roll out across 150 markets, including the US, European Union (EU) and Hong Kong, Bloomberg reported.
Subscribe to our newsletter
Get breaking news, exclusive insight, and expert analysis - before anyone else.
The hypertension detection feature will be supported on Apple Watch Series 9, 10, and 11, as well as Ultra 2 and Ultra 3 models.
According to Apple, hypertension affects roughly 1.3 billion adults worldwide and remains a major risk factor for heart disease, stroke and kidney complications. The iPhone-maker noted the tool was developed using advanced machine learning and trained on datasets from studies involving more than 100,000 participants.
The feature underscores Apple’s continued expansion into wearable health technology as it contends with rising competition from companies including Samsung and Oura Health.
Subscribe to our newsletter
Get breaking news, exclusive insight, and expert analysis - before anyone else.





Comments